Founded in 2012, Vdi Medical is the first domestic medical device manufacturer to have its independently developed AED (Automated External Defibrillator) products certified by NMPA. It offers a full range of AEDs and pre-hospital emergency solutions. With a commitment to "Life First," our factory integrates advanced AI diagnostic algorithms with robust hardware engineering.
Vdi Medical AED deployments have reached nearly 100,000 units, securing the second-highest domestic market share, and its products have entered over 30 countries and regions. Our AEDs demonstrated a first-shock success rate exceeding 98% in animal trials and have successfully resuscitated over 150 patients to date.
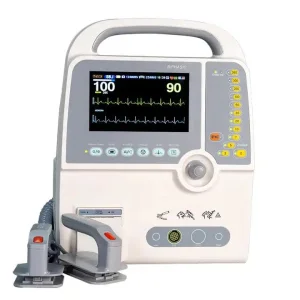
200+
Lives saved by Vdi Medical AEDs
98%+
First-shock success rate
100,000
AEDs installed globally
300,000
Annual production capacity
The global demand for Hospital-Grade AEDs is experiencing unprecedented growth. Driven by stringent workplace safety regulations and a rising awareness of Public Access Defibrillation (PAD) programs, industries from oil and gas to logistics are prioritizing emergency cardiac care. As a leading AED supplier, we recognize that "Hospital-Grade" is no longer confined to clinical wards—it is the new standard for corporate and public safety.
Modern AED technology is shifting toward IoT integration and AI-assisted diagnostics. Remote monitoring of AED readiness—battery status, pad expiration, and environmental conditions—allows large-scale corporate fleets to manage thousands of devices through a single cloud dashboard. Furthermore, the push for "Smart Cities" has integrated AEDs into public infrastructure, making them as ubiquitous as fire extinguishers.
Procurement officers for government agencies, educational institutions, and healthcare groups face a critical challenge: balancing clinical reliability with ease of use. A hospital-grade AED must be sophisticated enough for a paramedic yet intuitive enough for a teacher or a factory worker. Localization is key—this includes not only language prompts but also adherence to regional resuscitation guidelines (AHA vs. ERC).
As a China-based AED factory, Vdi Medical provides the most competitive "cost-per-life-saved" ratio. We offer OEM/ODM services that allow local brands to integrate our proven 98% first-shock success technology into their own market-specific housing and software.
Why do global medical distributors choose us as their AED supplier? It’s our fusion of clinical excellence and manufacturing efficiency. With an annual capacity of 300,000 units, we are prepared for the world's largest tenders while maintaining the meticulous quality control required for life-saving medical devices.
NMPA certified with proprietary algorithms that ensure accurate shock delivery, minimizing tissue damage and maximizing resuscitation probability.
ISO13485 and CE approved production lines. Every unit undergoes rigorous 100% testing before leaving our facility.
Proven track record in 30+ countries, with support for localized voice prompts, manuals, and regulatory documentation.