Advanced cardiac resuscitation devices designed for rapid response in public and professional French environments.

Sy-C025 First Aid Medical Portable Automated External Defibrillator AED (France Special Edition)

Sy-C025 First Aid Medical Portable Automated External Defibrillator AED

Portable AED Defibrillator Automated External Defibrillator AED with CE Sy-C025p

Wap A180 Medical Machine Biphasic Monitor Portatil Desfibrilador Portable Defibrillator AED
France has long been a pioneer in public health initiatives, particularly regarding Out-of-Hospital Cardiac Arrest (OHCA). As a leading Portable AED Manufacturer and Exporter in France, Vdi Medical recognizes the stringent regulatory environment governed by ANSM (Agence nationale de sécurité du médicament et des produits de santé). The French "Plan National de Santé" emphasizes the widespread deployment of Automated External Defibrillators in public spaces, creating a robust demand for highly reliable, portable technology.
In recent years, the French market has shifted towards "Smart AEDs"—devices integrated with IoT capabilities for remote monitoring. This ensures that every portable AED located in the Paris Metro, at the Eiffel Tower, or in remote villages of Provence is always ready for use. Our export operations to France focus on providing bilingual (French/English) voice prompts and adhering to the latest ERC (European Resuscitation Council) guidelines.
🚀
Technological Trends: The convergence of AI and emergency medicine is defining the next generation of defibrillators. In France, there is a growing trend of integrating AED data with the "SAU" (Services d'Accueil des Urgences) systems, allowing first responders to receive real-time data from the portable unit before they even arrive on the scene.
Local Applications: From the high-speed TGV trains to the corporate offices in La Défense, portable AEDs are becoming mandatory safety equipment. French law (Loi n° 2018-515) now requires many public-access buildings (ERP) to be equipped with these life-saving devices, further accelerating the need for high-quality manufacturing and reliable export partners.
Founded in 2012, Vdi Medical is the first domestic medical device manufacturer to have its independently developed AED (Automated External Defibrillator) products certified by NMPA. It offers a full range of AEDs and pre-hospital emergency solutions.
Vdi Medical AED deployments have reached nearly 100,000 units, securing the second-highest domestic market share, and its products have entered over 30 countries and regions. Vdi Medical AEDs demonstrated a first-shock success rate exceeding 98% in animal trials and have successfully resuscitated over 150 patients to date.
Read More »Lives saved globally
First-shock success
Units Installed
Annual Capacity
Optimized solutions for the diverse landscape of French infrastructure.






Entering the French medical device sector requires more than just a CE mark. It requires a commitment to quality and post-market surveillance. As a Portable AED Exporter, Vdi Medical provides comprehensive technical support to French distributors. Our devices feature advanced biphasic waveforms that adjust impedance based on the patient's body type—a crucial feature for the diverse demographic of France.
The French industrial sector, including automotive and aerospace hubs in Toulouse and Lyon, increasingly incorporates AEDs into their Health and Safety (HSE) protocols. Our products are designed to withstand rugged industrial environments, with high IP ratings (Ingress Protection) that protect against dust and water—ideal for both the Atlantic coast and the industrial northeast.
🛡️
Reliability and Maintenance: One of the major challenges in France is the maintenance of deployed AEDs. Our portable units feature automated self-tests and status indicators that are easily understood by non-medical personnel, ensuring the device is "Ready to Rescue" at any given moment.
Available for immediate distribution across all French regions and overseas territories.
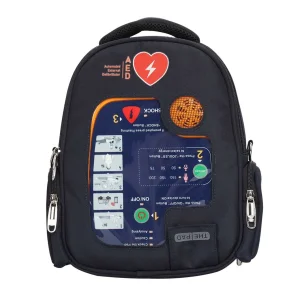
Portable Biphasic CPR Machine Cardiac Automated External Defibrillator AED

Mindray BeneHeart S1 Portable Automatic External Defibrillators (AED)

Mini Defibrillator Medical Automated External Defibrillator Portable AED
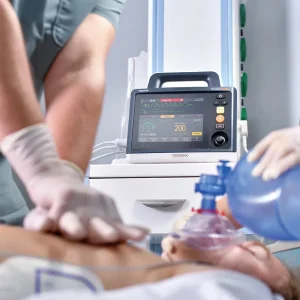
Mindray Beneheart D60 Automated External Portable AED Defibrillator

Cmics Dp1 Medical First Aid Portable Automated External Defibrillator AED

Qingdao Portable Cardiac Monitor Analyzer Automatic External Defibrillator AED

Karestar K-Ad20 Cardiac Monitor Portable Manual Defibrillator for Ambulance AED