 Automatic External Defibrillator (AED) with Low Energy, Adult/Pediatric Compatible Pads, Bluetooth
Automatic External Defibrillator (AED) with Low Energy, Adult/Pediatric Compatible Pads, Bluetooth
 AED with Low Energy, Adult/Pediatric Compatible Pads, Bluetooth
AED with Low Energy, Adult/Pediatric Compatible Pads, Bluetooth
 High-Precision AED with Universal Electrode Pads
High-Precision AED with Universal Electrode Pads
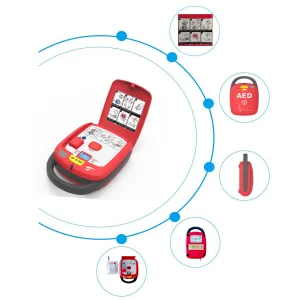 AED with Voice Guide, Bluetooth, Adult/ Pediatric Compatible Pads
AED with Voice Guide, Bluetooth, Adult/ Pediatric Compatible Pads
 Cardiology Defibrillator with High Capacity Battery & Preinstalled Pads
Cardiology Defibrillator with High Capacity Battery & Preinstalled Pads
 AED with Automatic Recording & Adult/Pediatric Pads
AED with Automatic Recording & Adult/Pediatric Pads
 Professional Adults/Children Defibrillator Training Electrode
Professional Adults/Children Defibrillator Training Electrode
 CE/ISO13485 Certified AED with Compatible Pads
CE/ISO13485 Certified AED with Compatible Pads
The global medical landscape is witnessing a paradigm shift in emergency cardiac care. Universal Electrode Pads have emerged as a critical component in the chain of survival for Sudden Cardiac Arrest (SCA). Unlike traditional model-specific accessories, universal pads offer a streamlined solution for diverse AED brands, reducing the logistical burden for large-scale facilities and medical distributors.
Currently, the market valuation for AED accessories is projected to grow at a CAGR of over 8% through 2030. This growth is driven by increasing government mandates for public-access defibrillation (PAD) programs in North America, Europe, and the Asia-Pacific region. As a result, the demand for high-quality, long-shelf-life, and cost-effective electrode pads has skyrocketed. China, as a global manufacturing powerhouse, has evolved from a basic supplier to a high-tech innovator in this field, with companies like Vdi Medical leading the charge with NMPA, CE, and ISO certifications.
In high-stress emergency environments, every second counts. Universal electrode pads are designed with proprietary hydrogel technology that ensures superior conductivity and skin adhesion across varied demographics—from pediatric to adult patients. This "one-size-fits-all" approach simplifies training for first responders and reduces the risk of error during pad replacement. For global procurement officers, this means lower inventory complexity and significant cost savings without compromising on clinical efficacy.
Technology is rapidly reshaping how electrode pads are designed and manufactured. Several key trends are defining the next decade of the industry:
International buyers—ranging from hospital chains to airport authorities—are no longer just looking for the lowest price. The modern procurement checklist includes regulatory compliance (FDA/CE), shelf-life stability (ideally 2-3 years), and supply chain resilience. Chinese exporters have adapted by offering "white-glove" OEM/ODM services, allowing global brands to customize electrode pad designs while leveraging the high-efficiency production lines found in hubs like Shenzhen and Suzhou.
Founded in 2012, Vdi Medical is the first domestic medical device manufacturer to have its independently developed AED (Automated External Defibrillator) products certified by NMPA. It offers a full range of AEDs and pre-hospital emergency solutions.
Vdi Medical AED deployments have reached nearly 100,000 units, securing the second-highest domestic market share, and its products have entered over 30 countries and regions. Our AEDs demonstrated a first-shock success rate exceeding 98% in animal trials and have successfully resuscitated over 150 patients to date.
Lives Saved Globally
First-Shock Success
AEDs Installed
Annual Capacity
When sourcing from the top 10 Chinese suppliers, global exporters benefit from a unique combination of scale, speed, and sophistication:
 First Aid AED Cardiac Defibrillator with Preinstalled Pads
First Aid AED Cardiac Defibrillator with Preinstalled Pads
 Emergency First Aid Defibrillator with High-Stability Pads
Emergency First Aid Defibrillator with High-Stability Pads
 Professional Cardiac Defibrillator (AED) Solutions
Professional Cardiac Defibrillator (AED) Solutions
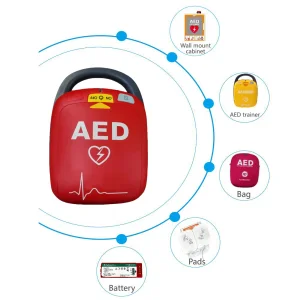 CE Certified AED with Low Energy BTE Technology
CE Certified AED with Low Energy BTE Technology
 Competitive AED with Universal Adult/Pediatric Pads
Competitive AED with Universal Adult/Pediatric Pads
 AED with Customized Multi-Language Support
AED with Customized Multi-Language Support
 BTE Technology AED with Universal Compatible Pads
BTE Technology AED with Universal Compatible Pads
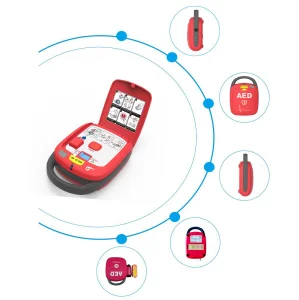 Bluetooth Integrated AED with Smart Electrode Pads
Bluetooth Integrated AED with Smart Electrode Pads