The global market for Adult Defibrillation Pads is currently experiencing an unprecedented surge, driven by increasing awareness of public health safety and the rising incidence of sudden cardiac arrest (SCA) worldwide. Sudden cardiac arrest remains one of the leading causes of death globally, necessitating the widespread installation of Automated External Defibrillators (AEDs) in public spaces, corporate environments, and medical facilities. As the critical interface between the AED device and the patient, defibrillation pads are the most frequently replaced consumable in the emergency response ecosystem.
Industrial data indicates that the market is transitioning from traditional hospital-based defibrillation to decentralized public access defibrillation (PAD) programs. This shift has created a massive demand for high-quality, long-shelf-life adult defibrillation pads. In North America and Europe, stringent regulatory standards such as the MDR (Medical Device Regulation) and FDA pre-market approvals have raised the bar for manufacturers, ensuring that only the most reliable electrodes reach the market. Meanwhile, in emerging markets across Asia and the Middle East, government-led initiatives are mandating AED installations in airports, subways, and sports stadiums, further fueling the demand for wholesale suppliers and exporters from manufacturing hubs like China.
Today's industrial landscape is not just about manufacturing a simple electrode; it's about the precision of the conductive hydrogel, the shelf-life stability under various climatic conditions, and the universal compatibility with major AED brands. As a result, global procurement managers are increasingly looking for partners who can offer a balance of technological innovation, regulatory compliance, and cost-efficiency.

Our premium defibrillation pads are designed for rapid deployment. Featuring advanced conductive hydrogel for superior skin adhesion and optimized energy delivery. These pads are compatible with multiple AED models and meet all international safety standards.
View Product DetailsThe defibrillation industry is undergoing a digital and material science revolution. One of the most significant trends is the integration of "Smart" technology into defibrillation pads. Modern electrodes are now being developed with sensors that provide real-time feedback on CPR quality, including compression depth and rate, allowing the AED to guide the rescuer more effectively. This synergy between the pad and the software is significantly increasing survival rates.
Another emerging trend is the move toward "Universal Pads." Traditionally, rescuers had to switch between adult and pediatric pads, which could waste precious seconds during a cardiac emergency. The latest industry standard involves a single set of pads compatible with both adults and children, where the energy levels are adjusted via a switch on the AED itself. This simplifies the user experience and reduces inventory costs for organizations.
Furthermore, sustainability is becoming a key focus. While pads are inherently single-use consumables, manufacturers are exploring biodegradable materials and more eco-friendly packaging to reduce the environmental footprint of the hundreds of millions of pads produced annually. As a leading China exporter, we are at the forefront of these trends, investing heavily in R&D to ensure our adult defibrillation pads are not only life-saving but also technologically advanced and future-proof.
Procuring adult defibrillation pads on a global scale involves navigating a complex web of technical specifications and regulatory requirements. Professional buyers—ranging from hospital groups and government health ministries to large-scale private corporations—prioritize three main factors: Reliability, Compatibility, and Shelf-Life.
Reliability: In a life-or-death situation, the failure of an electrode is not an option. Buyers require rigorous clinical data and third-party certifications (such as CE, ISO 13485, and NMPA) to verify that the pads will deliver the necessary shock effectively through various skin types and conditions.
Compatibility: With various AED brands like Philips, Zoll, Physio-Control, and our own Vdi Medical units on the market, wholesale buyers seek suppliers who can provide cross-compatible solutions or reliable OEM alternatives that function perfectly with existing hardware.
Shelf-Life: Since AED pads are "standby" items, they must remain viable for 2 to 5 years. Procurement teams look for advanced foil-packaging technology that prevents the hydrogel from drying out, ensuring the pads are ready for use even after years of storage.
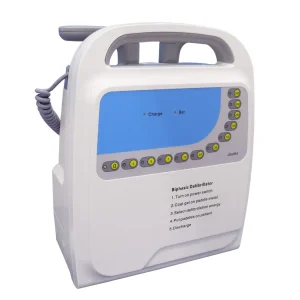
Designed for professional medical environments, this biphasic defibrillator offers manual, asynchronous, and synchronized modes. It works seamlessly with our high-conductivity adult pads to provide reliable life support in critical care settings.
Learn MoreThe application of adult defibrillation pads is no longer confined to the back of an ambulance. Localization has led to specific designs for varied environments. In the industrial sector, pads are designed to be rugged and resistant to high-vibration environments like oil rigs or mining sites. In the public sector, the focus is on intuitive visual instructions printed directly on the pads to assist untrained bystanders.
In high-traffic areas such as international airports and transit hubs, pads are often stored in outdoor cabinets where they must withstand extreme temperature fluctuations. Our manufacturing process includes environmental stress testing to ensure the hydrogel remains stable from -30°C to +65°C. This level of specialization allows our pads to be deployed successfully from the humid tropics of Southeast Asia to the cold climates of Northern Europe.






Founded in 2012, Vdi Medical is the first domestic medical device manufacturer to have its independently developed AED (Automated External Defibrillator) products certified by NMPA. It offers a full range of AEDs and pre-hospital emergency solutions.
Vdi Medical AED deployments have reached nearly 100,000 units, securing the second-highest domestic market share, and its products have entered over 30 countries and regions. Vdi Medical AEDs demonstrated a first-shock success rate exceeding 98% in animal trials and have successfully resuscitated over 150 patients to date.
As a specialized China factory, we leverage an integrated supply chain that allows us to provide wholesale adult defibrillation pads at highly competitive prices without compromising on medical standards. Our facility is equipped with state-of-the-art automated production lines capable of producing 300,000 units annually. This scale ensures that we can meet the large-volume requirements of international exporters and government tenders with short lead times.
Our R&D team continuously works on improving the polymer science behind our hydrogels, ensuring lower impedance and higher energy efficiency. By choosing us as your supplier, you are partnering with a company that combines the agility of Chinese manufacturing with the rigorous quality control of a global medical leader.
Lives Saved Globally
First-Shock Success Rate
Units Installed Worldwide
Annual Production Capacity
The effectiveness of a defibrillation shock depends largely on the "Impedance" or the resistance between the pads and the patient's heart. Our adult defibrillation pads are engineered with high-purity metal foil layers and a proprietary hydrogel formula that minimizes skin-electrode impedance. This ensures that the maximum amount of energy reaches the myocardium, increasing the likelihood of successful defibrillation on the first attempt.
Furthermore, we address the common issue of "skin burns" by ensuring even current distribution across the entire surface of the pad. Many low-quality pads suffer from "edge effects" where the current concentrates on the borders of the electrode; our advanced manufacturing process eliminates this risk. The lead wires are also designed with high-flexibility insulation to prevent kinking or breaking during the high-stress environment of a rescue operation.
For wholesale partners, we provide customizable packaging options. Whether you need bulk shipping for hospital systems or individual retail-ready kits for consumer AED sales, our packaging is designed to maintain the sterile integrity and moisture level of the gel for a minimum of 36 months, even in non-climate-controlled environments.
As we look to the future, Vdi Medical is pioneering the use of AI-driven manufacturing analytics to monitor every single pad that comes off our production line. By analyzing the consistency of the hydrogel thickness and the conductivity of the leads in real-time, we maintain a defect rate that is among the lowest in the industry. This commitment to "AI Tech" in our production process is what sets us apart as the premier China supplier and exporter.