Founded in 2012, Vdi Medical is China's first NMPA-certified independent AED manufacturer — delivering world-class automated external defibrillators and pre-hospital emergency solutions to over 30 countries and regions worldwide.
Founded in 2012, Vdi Medical is the first domestic medical device manufacturer to have its independently developed AED (Automated External Defibrillator) products certified by NMPA. It offers a full range of AEDs and pre-hospital emergency solutions.
Vdi Medical AED deployments have reached nearly 100,000 units, securing the second-highest domestic market share, and its products have entered over 30 countries and regions.
Vdi Medical AEDs demonstrated a first-shock success rate exceeding 98% in animal trials and have successfully resuscitated over 150 patients to date.
Backed by over a decade of R&D, clinical validation, and global deployment experience, Vdi Medical delivers unmatched performance and reliability.
In-house engineering team with full IP ownership over AED algorithms, hardware design, and firmware — enabling rapid iteration and customization.
A first-shock defibrillation success rate exceeding 98% in controlled animal trials, with over 150 confirmed real-world patient resuscitations.
Products certified under NMPA, CE, FDA, and other major international standards, enabling seamless entry into regulated markets worldwide.
Annual production capacity of 300,000 AED units with ISO 13485-certified quality management systems ensuring consistent product excellence.
Comprehensive white-label and custom-design services for global distributors, with full support from concept to delivery and post-sale training.
Next-generation AEDs with real-time cloud monitoring, remote diagnostics, and integration with emergency dispatch systems for faster response.
From government health agencies to international distributors, organizations worldwide trust Vdi Medical for life-saving technology.
China's first NMPA-certified independent AED developer — a heritage of regulatory firsts and technical breakthroughs.
Factory-direct pricing with no middlemen, making high-performance AEDs accessible for public deployment at scale.
Streamlined supply chain with fast lead times and global shipping partnerships to meet urgent deployment needs.
Comprehensive after-sales service including training programs, maintenance contracts, and 24/7 technical support.
Vdi Medical's AEDs meet the most stringent international regulatory requirements, giving you confidence in every deployment.
First independently developed AED to receive National Medical Products Administration approval in China — the gold standard of domestic compliance.
ChinaFull compliance with EU Medical Device Regulation, enabling legal sale and distribution across all 27 European Union member states.
EuropeUS Food and Drug Administration clearance confirms safety and effectiveness equivalence to legally marketed AED devices in the United States.
USAInternational standard for medical device quality management systems — ensuring consistent manufacturing processes and traceability.
InternationalElectrical safety and essential performance standards for medical electrical equipment — verified through independent third-party testing.
Safety StandardRegulatory approvals spanning Southeast Asia, Middle East, Latin America and Africa — supporting deployment in 30+ countries and regions.
GlobalVdi Medical is a preferred OEM partner for leading global medical device brands and distributors. Our vertically integrated manufacturing facility supports every stage of product development — from industrial design and firmware engineering to regulatory filing and logistics.
With an annual capacity of 300,000 AED units, we deliver consistent quality at scale, backed by ISO 13485-certified processes and a dedicated OEM project management team.
Vdi Medical provides tailored AED solutions and pre-hospital emergency programs for a wide range of public and private settings.

Shopping malls, airports, stadiums, and community centers — high-traffic environments where rapid AED access is critical.
High Priority
Athletes and active individuals face elevated cardiac risk — Vdi Medical AEDs are built for fast, confident response in sports environments.
Sports Safety
Compact, easy-to-use AEDs designed for households with elderly or at-risk family members — life-saving technology within reach.
Personal Safety
Protecting students, staff, and visitors with AEDs designed for non-medical responders — simple voice guidance for confident use by anyone.
Education Sector
Subway stations, train carriages, airports, and bus terminals — strategic AED placement for dense passenger environments.
Transit Safety
Corporate workplaces and business parks equipped with AEDs and employee training programs to build a heart-safe work culture.
Workplace SafetyVdi Medical AEDs have been deployed in landmark public safety programs across China and internationally — here are some of our most impactful projects.
Deployed 5,000+ Vdi Medical AEDs across subway stations, parks, and public buildings as part of a city-wide cardiac emergency response initiative, reducing emergency response time by over 40%.
Supplied and installed AEDs throughout a major international airport hub serving 40 million annual passengers, with staff training and IoT-based device monitoring integrated into the airport's emergency system.
Partnered with provincial education bureaus to equip thousands of primary and secondary schools with AEDs and CPR training programs, creating a new standard for campus health safety.
Comprehensive AED deployment for professional sports venues and fitness chains, including integration with venue management systems and regular device health checks via cloud dashboard.
Manufactured and supplied 10,000+ white-label AED units for a major European medical device distributor, including full CE documentation, custom packaging, and multi-language user guides.
Equipped an entire urban metro system with strategically placed AEDs at every station, integrated with the city's 120 emergency dispatch center for real-time incident response coordination.
Vdi Medical's advanced production campus is purpose-built for precision medical device manufacturing, combining cutting-edge automation with rigorous quality control.
Purpose-built GMP-compliant manufacturing campus with dedicated clean rooms and assembly lines.
High-precision surface-mount technology production lines ensuring consistent PCB assembly quality at scale.
Dedicated biomedical engineering laboratory for algorithm development, hardware prototyping, and clinical validation testing.
100% electrical safety testing, defibrillation output verification, and environmental stress screening on every unit before shipment.
Hear from the hospitals, distributors, and public safety organizations that rely on Vdi Medical every day.
"We have deployed Vdi Medical AEDs across 120 subway stations in our city. The device reliability is outstanding, and the IoT monitoring platform has transformed how we manage our AED network. Response times have improved dramatically."
"As a European distributor, finding a reliable OEM partner was critical. Vdi Medical exceeded all our expectations — from CE documentation to custom packaging and on-time delivery. Our customers love the product quality."
"We installed Vdi Medical AEDs in all our fitness centers nationwide. The simple voice-guided interface means any staff member can respond confidently. We've already had one successful resuscitation — this product saves lives."
Contact Vdi Medical today to discuss your AED deployment project, OEM partnership, or distribution inquiry. Our team is ready to support you.
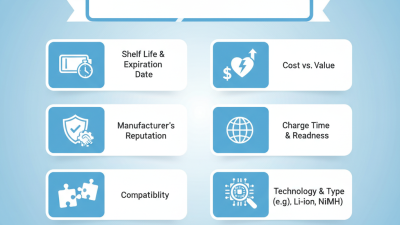
When purchasing a replacement AED battery, careful consideration of various factors is crucial. Mark Johnson, an expert in emergency medical
Author: Madeline Date: 2026-03-30
Learn More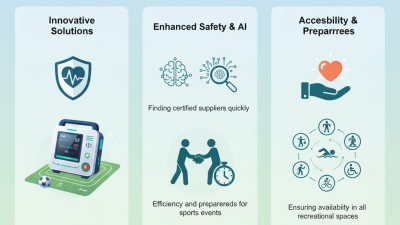
The 2026 Canton Fair promises to be a groundbreaking event, showcasing innovative solutions, including Sports Field AEDs. With the increasing demand
Author: Ethan Date: 2026-03-27
Learn More
At the upcoming China Import and Export Fair, various innovations, including Short Cycle AEDs, will take center stage. The demand for advanced
Author: Charlotte Date: 2026-03-25
Learn More